Chem: Structure of pramanu(atom)
help please tumhara din accha ho jaye reason bhi dedena( with reason plz solve this question)
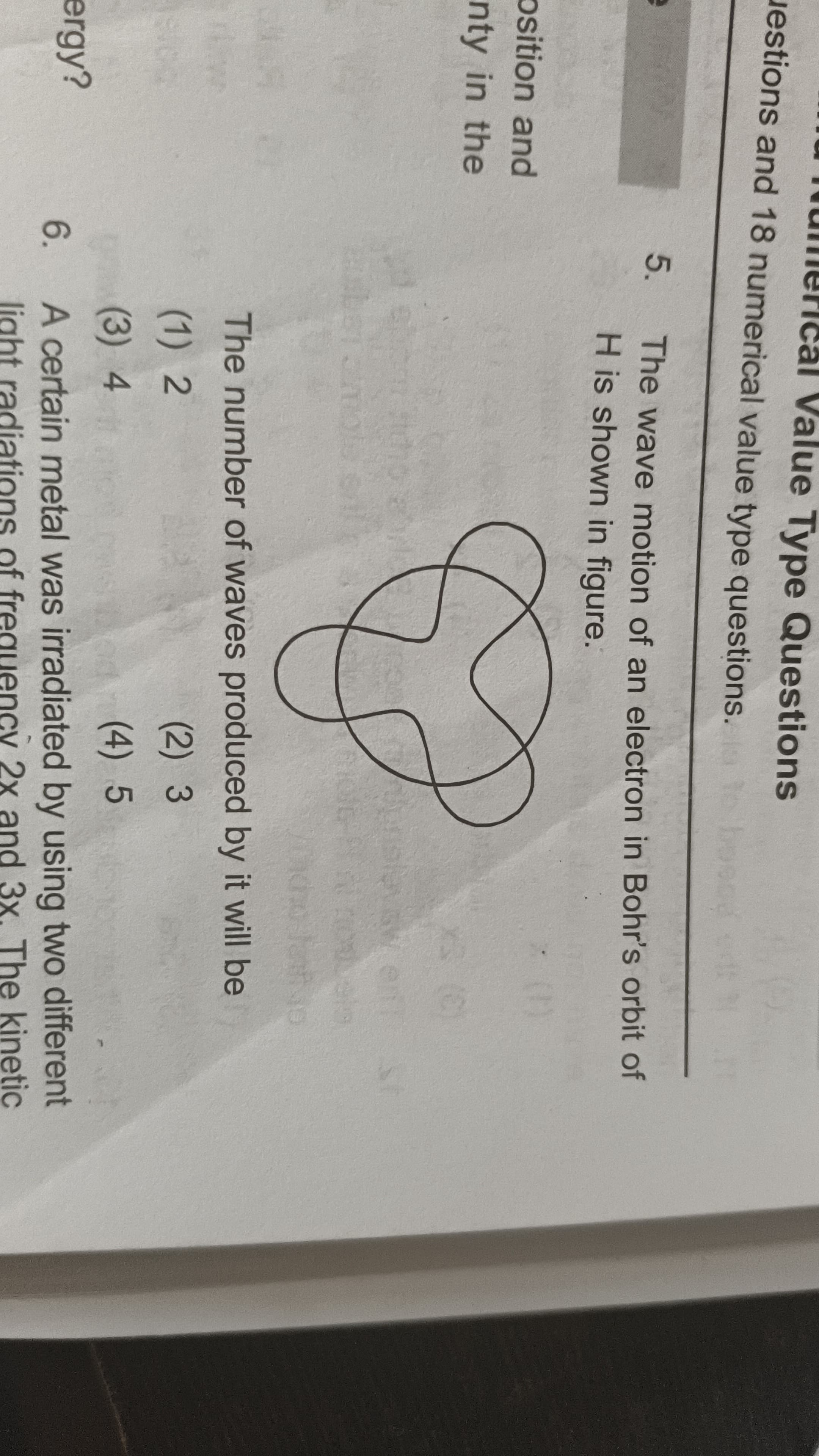
39 Replies
@Dexter
Note for OP
+solved @user1 @user2... to close the thread when your doubt is solved. Mention the users who helped you solve the doubt. This will be added to their stats.,rotate

what
rotate kya
yus ho jaata hai
uhh this is a topic taught in 12th generally (i think)
is the answer 3?
bilkul 3 hi hai
haan so basically

,rotate

yes that basically
Bohr said that mvr = nh/2pi
for atomic model
an electron revolves in that circular orbit in which its circumference is an integral multiple of its de broglie wavelength
to justify that, Debroglie showed this wave nature of an electron
if you want more context behind that
yes
look up 12th ncert Dual Nature chapter
batao bhai deep mai chem ka alag hi feel hai
ill tell you the subtopic name hang on
reallll
mil nhi raha aise hi bata deta hoon
so basically
Bohr said that for atomic model
mvr = nh / 2pi
but he didnt know why
now Debroglie had said that all matter also has wave nature (debroglie wavelength)
toh unhone bola ki it happens since electrons exist as standing waves in the orbit
standing wave matlab the type of wave that goes up and down only, like when you pluck a guitar string
that flower like shape is just shown to represent a wave
3 toh integral multiple nahi hai
as u can see there are 3 crests, so three waves
3 integer hi toh hai 😭
n=3 here
I really admire de Broglie's guts for proposing this ngl
oo
literally the perfect advertisement for his matter wave theory
He wasn't exactly right, Schrödinger was the one who perfected his argument, but by God, who thinks of this?
Ah yes we have tiny charged spheres in orbits. But they're actually not particles, they're waves.
nimboi mai thoda chem mai dhila tha lekin akhri mai maza aaraha hai
how the hell did bohr think of mvr = nh/2pi with no debroglie to back it up
yesss (this is more physics than chem but acha laga sun ke)
Amazing quote from the QM series i watched.
"Physics is not coming up with a theorem and proving it. We leave that to the mathematicians. We construct a model. If it fits the data, we use it. If it doesn't ,we toss it and think up a new one."
You do not derive these. You make it up, and check against data.
tum sabka din accha ho👍
Like Rydberg's formula for Balmer's spectrum. It is purely empirical.
Bohr provided a theoretical explanation which matched the observation.
And from that he got the angular momentum quantization
wow
physics is very creative in that sense
wHAT
insane
the Q and the learning it gave👏
I like how we all just discuss things irl WhatsApp and discord collectively now Internet OP
bro made chatgpt explain it 😭
indians bhi toh job outsource kar sakte hain 😤
BPO hiring another BPO to BPO
+solved @Nimboi
Post locked and archived successfully!
Archived by
<@1035556259417571408> (1035556259417571408)
Time
<t:1742859771:R>
Solved by
<@717724055217635398> (717724055217635398)