Chemical Bonding
I don't think they're all 0 cause well this is a JEE question xd. I saw something online saying O--H bond is rotatable so dipole moment is non-zero but I dont understand it fully

30 Replies
@Dexter
Note for OP
+solved @user1 @user2... to close the thread when your doubt is solved. Mention the users who helped you solve the doubt. This will be added to their stats.nope its not
3 and 4 have mu not equal to 0
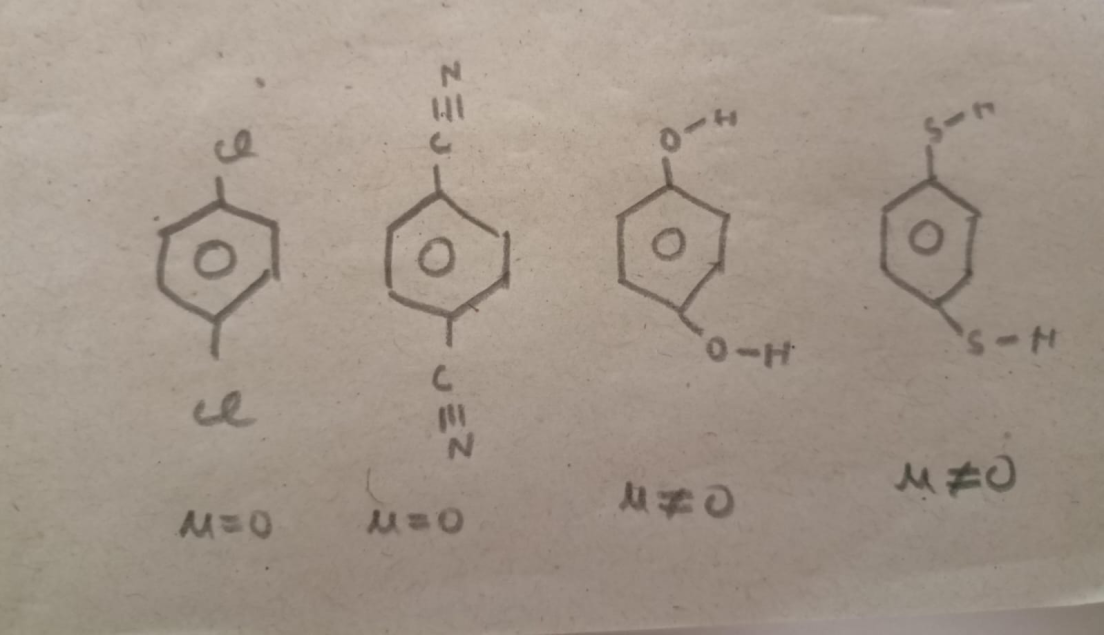
Thanks a lot
Thanks sir. So can I say that 1,3 dichloro propane/ 1,4-butanediol etc have non-zero dipole moment cause of lone pair on O and Cl atoms?
And though this is a PYQ im not sure what the full quesiton was, but is it possible to find which has more Mu out of the non zero dipole moment compds?
vector sum
Oh yeah
The OH and SH have bent geometry
Yes due to LP right
yeap
@Dexter Can you tell if I'm right?
hmm interesting
see
agar kisi bhi confirmation mein dipole moment zero aajaye
then us compound ka dipole moment zero hi kehte hai
yeah that seems to check out
maybe in compounds mein kisi conformer mein dipole moment zero ho
yeap, 1,3-dichloropropane has a dipole moment of 2.09D
yeah
they still keep rotating because of ambient environmental energy ryt
they dont stay in the most stable conformer
yeah
thats why conformers exist
hmm yes
so yeah nonzero dipole
Its the same as optical activity
agar kisi bhi conformer mein optically inactive hogaya compound then whole compound is rendered optically active
Ahh so only in a particular confirmation of 1,3 dichloropropane and only for a short time dipole moment is zero/
yeah, so basically never since they keep rotating
Hmm yes thank you
Shall I mark sovled
yus tumhari marzi
+solved @burrito @iTeachChem @Dexter @Nimboi
Post locked and archived successfully!
Archived by
<@978554723454042192> (978554723454042192)
Time
<t:1741162399:R>
Solved by
<@1077867258631688192> (1077867258631688192), <@1035556259417571408> (1035556259417571408), <@1095655769376034818> (1095655769376034818), <@759051317124792351> (759051317124792351)